Coulometric Titrators and Volumetric Titrators
Karl Fischer Titration is a technique for the determination of moisture content. The technique was developed by a chemist named Karl Fischer. It is based on a reagent which reacts with water and converts the water into a non-conductive chemical. Karl Fischer provides for the specific detection of water content in a product.
There are two methods used to perform the Karl Fischer titration test.
One is known as Volumetric Karl Fischer. The other method is known as Coulometric Karl Fischer.
In this approach, the reagent and solvent are combined in the titration cell. When a sample is introduced into the titration cell and dissolved, reagent is released by the induction of an electrical current. The amount of current required to convert the water is the determinant of the amount of moisture. A Coulometric Karl Fischer instrument is often referred to as a coulometer.
The advantage of the Coulometric Karl Fischer method is the capability to accurately measure small amounts of moisture. Sensitivity of these instruments is as low as 0.1 microgram (µg) of water. This method is normally used for moisture content below 1% or for samples where the moisture is less than 200 micrograms.
 Range: 1 ppm to 100%
Range: 1 ppm to 100%
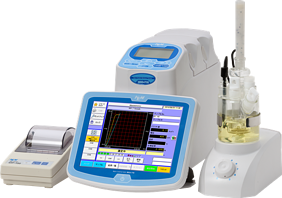

With this method, the moisture determination is based on the amount, or volume, of reagent used to convert the water. In the Volumetric Method, samples are dissolved in a solvent before the titration begins. A reagent is added until the water is removed.
VolumetricKarl Fischer is ideal when working with samples containing higher levels of moisture (generally over 1% or 2%) but also when samples may contain ketones and or aldehydes.
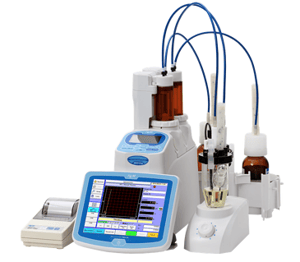 Measuring Range: Water Content 0.1 to 500mgH2O, Concentration 10ppm to 100%H2O
Measuring Range: Water Content 0.1 to 500mgH2O, Concentration 10ppm to 100%H2O
 For samples that cannot be dissolved in approved solvents and for samples that only release moisture at high temperatures, a Karl Fischer Oven-Evaporator is used. The evaporator removes the water, which is moved to the titration cell by a dry, inert gas, and the titration is carried out.
For samples that cannot be dissolved in approved solvents and for samples that only release moisture at high temperatures, a Karl Fischer Oven-Evaporator is used. The evaporator removes the water, which is moved to the titration cell by a dry, inert gas, and the titration is carried out.
© 2023 CSC Scientific Company, Inc. All rights reserved.