What seems like a million words ago, I began this journey explaining the simplest approach to measuring or determining moisture: Loss-on Drying. Little did I know how involved and esoteric it would become.
This voyage has taken me down  mysterious paths through spooky theories, back to age-old chemistry concepts and into the vagaries of thermodynamics related to evaporation, vapor pressure, bound water and water activity. I have come full-circle; back to explaining Loss-on Drying -- a form of drying that I had assumed would be the simplest of all.
mysterious paths through spooky theories, back to age-old chemistry concepts and into the vagaries of thermodynamics related to evaporation, vapor pressure, bound water and water activity. I have come full-circle; back to explaining Loss-on Drying -- a form of drying that I had assumed would be the simplest of all.
I thought the first four topics [evaporation, vapor pressure, bound moisture, water activity] were tough, complex, confounding and less-than-obvious. Drying -- defined as “the mass-transfer process of removing water (or other solute) by evaporation from a solid, semi solid or liquid” -- seemed easy.
As is often the case, reality makes “easy” a non-operative word. Such has turned out to be so with respect to the issue of drying.
To begin, there is the process in which heat is transferred to create a temperature in a solid that evaporates moisture from the surface and causes it to migrate from the inside outward. This internal migration occurs through several mechanisms such as diffusion, capillary action and the internal pressure created by shrinkage. It is not as simple as hanging laundry on a clothes line to dry on a breezy, sunny day.
This internal migration occurs through several mechanisms such as diffusion, capillary action and the internal pressure created by shrinkage. It is not as simple as hanging laundry on a clothes line to dry on a breezy, sunny day.
Firstly, there is the initial and underlying issue of product classification. Here are some examples:
-
Non-hygroscopic Capillary Porous Solids: These are materials such as sand, crushed minerals, certain crystals, polymer particles and some ceramics. They have recognizable pore space filled with liquid, little bound moisture and do not shrink on drying.
-
Hygroscopic Porous Solids:Clay, molecular sieves, wood, textiles, silica gels, alumina and zeolites are examples. They are characterized by clearly seen pore space, physically bound moisture and shrinkage in the early stages of drying.
-
Colloidal (non Porous) Solids: Examples include soap, glue, polymers like nylon and various food products. There is no pore space and all moisture -- except that on the surface -- is physically bound.
There are other less-common classifications and combinations as well.
In the drying process, the rate of moisture removal varies widely between these classifications. In some, the rate is linear throughout. In others, the removal-rate is high at the beginning and diminishes as drying progresses [as illustrated in the dreaded drying-rate curves]. The principles of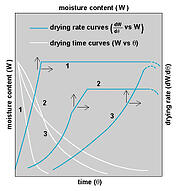 evaporation, equilibrium pressure, vapor pressure, partial pressure, temperature, relative humidity, dew point, moisture sorption isotherms and ideal gas laws all apply. As you may remember, some of these concepts have effect in other sections of the Moisture Mysteries Series. They all apply to this concluding phenomenon of drying, to which in the beginning I naively referred as just plain drying.
evaporation, equilibrium pressure, vapor pressure, partial pressure, temperature, relative humidity, dew point, moisture sorption isotherms and ideal gas laws all apply. As you may remember, some of these concepts have effect in other sections of the Moisture Mysteries Series. They all apply to this concluding phenomenon of drying, to which in the beginning I naively referred as just plain drying.
Most products from today's industry undergo drying at some stage. For example, drying is an important element in a wide range of products such a food, pharmaceuticals, lumber, paper, fiberboard and a host of chemical products. In fact, approximately 10% of the energy consumption in the US and Canada is directly related to production drying.
All of the issues that stretched my thought process are present with the development of production systems. Consequently, dryer designs are not universal across product classifications because of the inherent complexities in the drying process. Production drying systems are developed for specific products. These include convection, belt, fluid bed, rotary, spray, flash and drum dryers.
of the inherent complexities in the drying process. Production drying systems are developed for specific products. These include convection, belt, fluid bed, rotary, spray, flash and drum dryers.
One of the more difficult issues is to get at is the bound water. Among other techniques to deal with this is freeze-drying. Additionally, there are mechanical methods of removing moisture [e.g. centrifuging] that are referred to as dewatering rather than drying.
A compromise is often sought between the cost of transportation (lower moisture) and the cost of drying. Further, the level of drying is often a measure of a product’s suitability for market -- i.e. color, flavor, shrinkage, palatability and cracking. To check the effectiveness of these processes in meeting the diverse objectives, the moisture content after drying needs to be determined. The most frequently used technique of moisture determination is Loss-on Drying. That was the reason for this series of articles that sought to explain the factors of drying.
The Loss-on Drying moisture measurement process involves all issues and considerations concerned with production drying. People regularly ask us, “at what temperature should we run the test and for how long?” This discussion of drying should help them understand why we reply “It depends”.
I hope this “Loss-on Drying and Other Moisture Mysteries” series has been interesting and useful. Also I hope it was fun.
Now that I have finished Part V, the last in the series, I remain more astonished than ever with the mysteries of the test equipment world.
Thanks for visiting.
Art
P.S. Did you know that you can subscribe to these exposés, rants, raves and ramblings? To subscribe for email notice just enter your email address into the box just to the right of the title.
P.P.S. If you need to do LOD testing, take a look at why you might pick a CSC Digital Moisture Balance.

